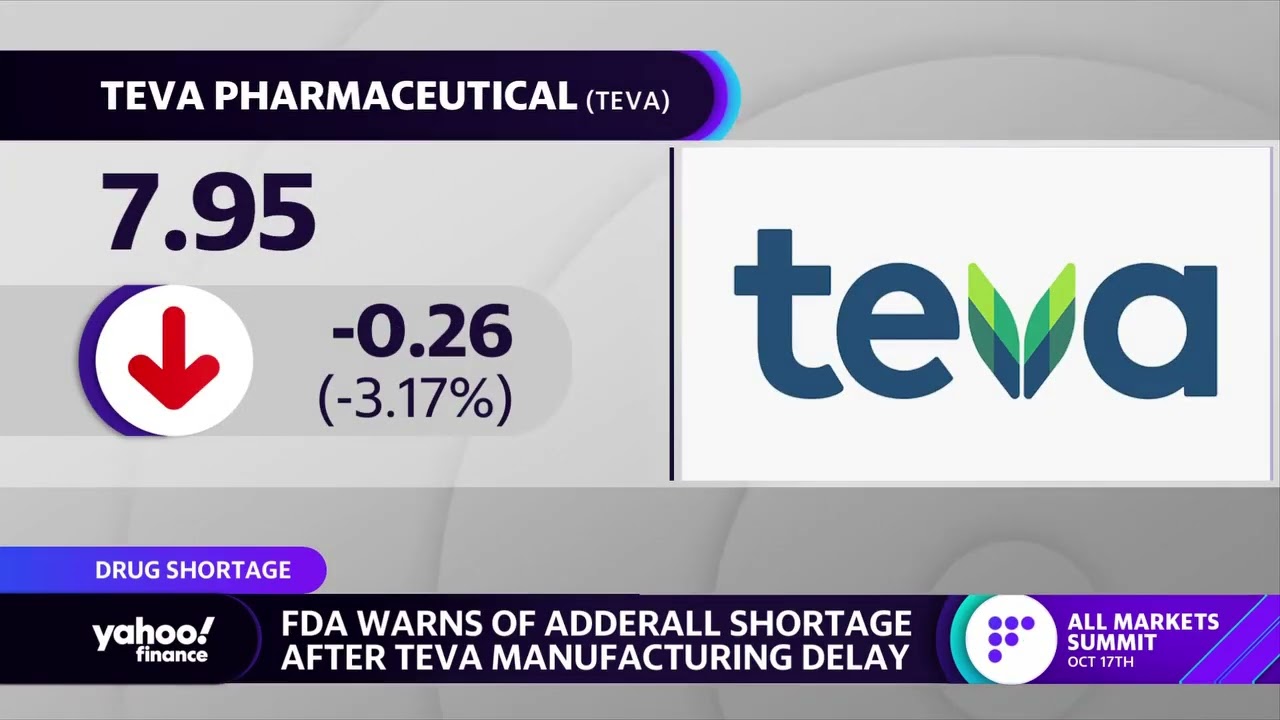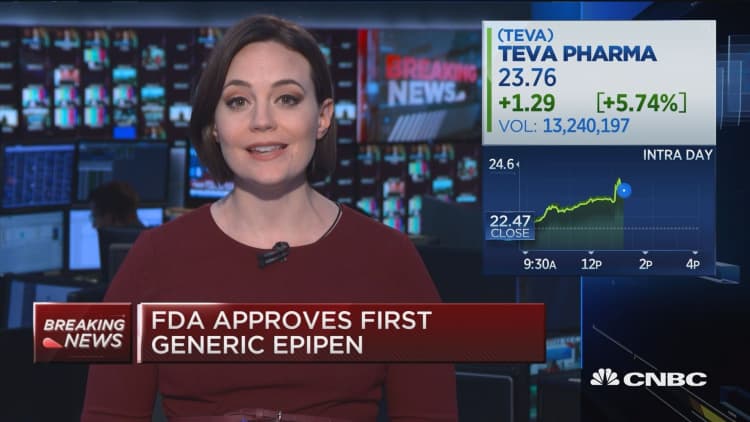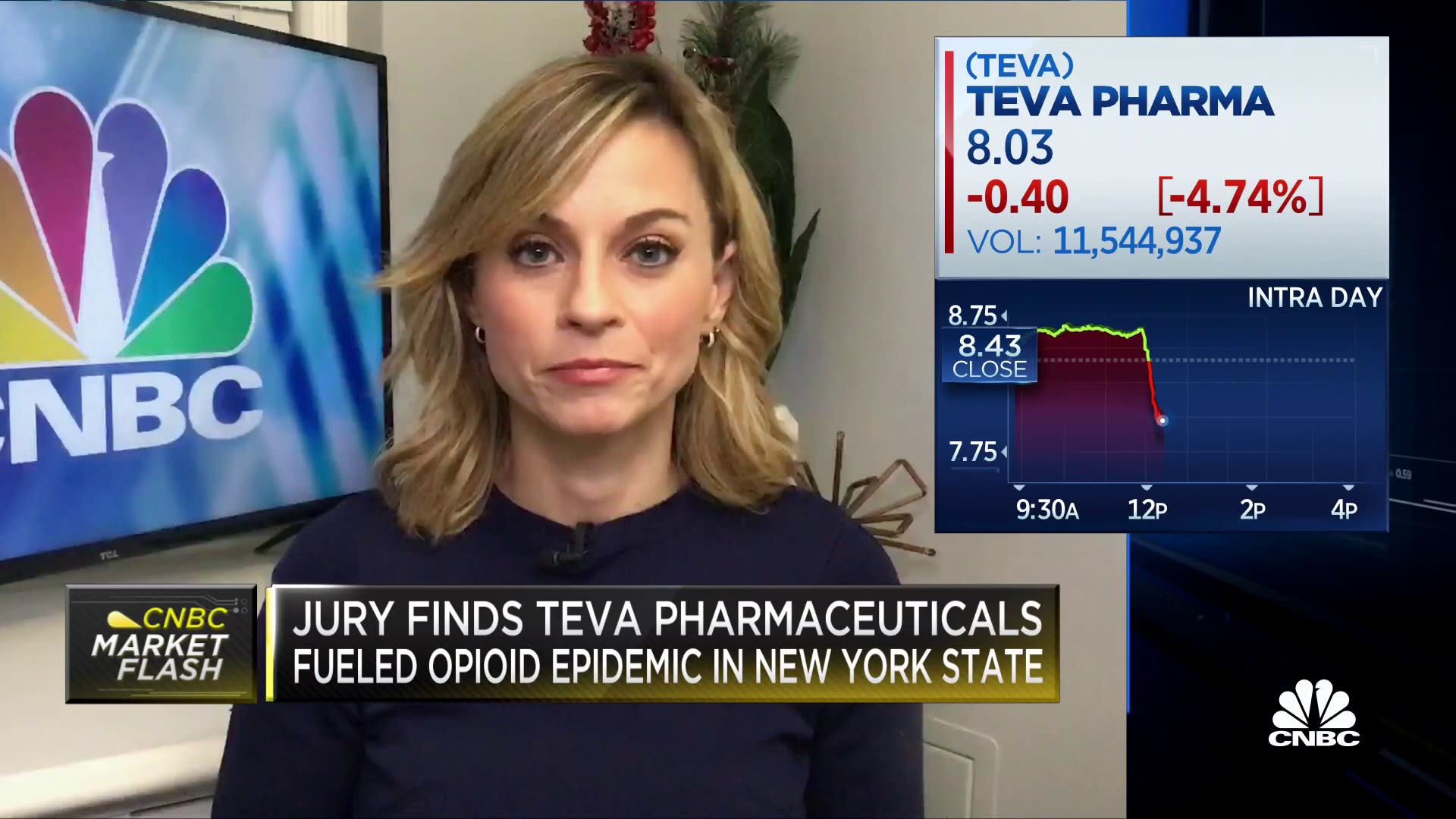
Teva, chasing Amgen's Aimovig, scores much-needed FDA approval for migraine drug Ajovy | Fierce Pharma

Teva Pharmaceuticals and MedinCell gain FDA approval for UZEDY, the first subcutaneous, long-acting formulation of risperidone for the treatment of schizophrenia in adults.

Teva Launches Two Digital Inhalers in the U.S., AirDuo® Digihaler® (fluticasone propionate and salmeterol) Inhalation Powder and ArmonAir® Digihaler® (fluticasone propionate) Inhalation Powder | Business Wire

Teva and MedinCell Announce FDA Approval of UZEDY™ (risperidone) Extended-Release Injectable Suspension, a Long-Acting Subcutaneous Atypical Antipsychotic Injection, for the Treatment of Schizophrenia in Adults | Business Wire

Teva Pharmaceuticals gets FDA approval for Osteoporosis injection | Eric A. Hughes, MD, PhD posted on the topic | LinkedIn