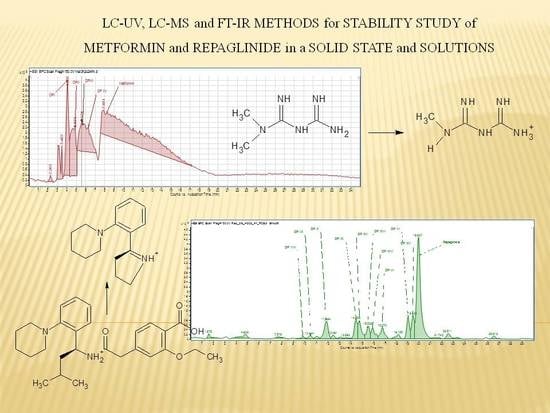
Bruxelles, le 21.11.2014 C(2014)9042 (final) DÉCISION D'EXÉCUTION DE LA COMMISSION du 21.11.2014 concernant le transfert de l
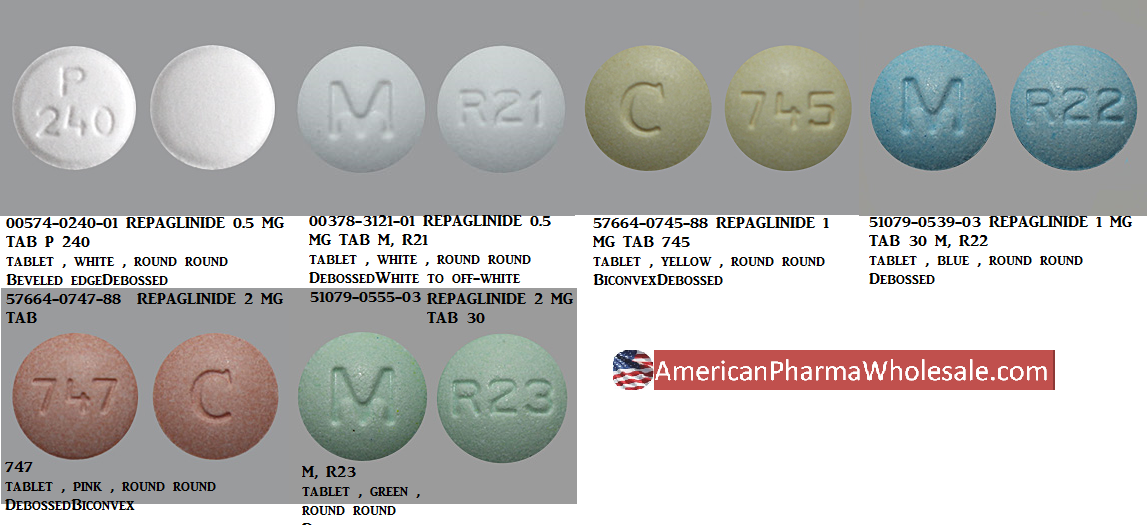
PRODUCT MONOGRAPH PrTEVA-REPAGLINIDE (repaglinide) 0.5 mg, 1 mg and 2 mg tablets Teva Standard Oral Antidiabetic Agent Teva Can