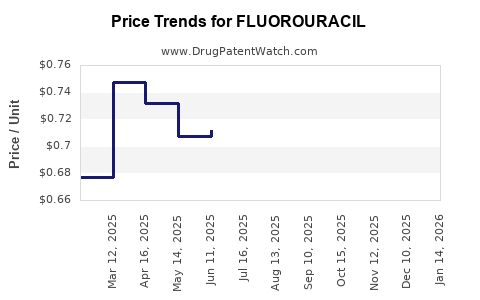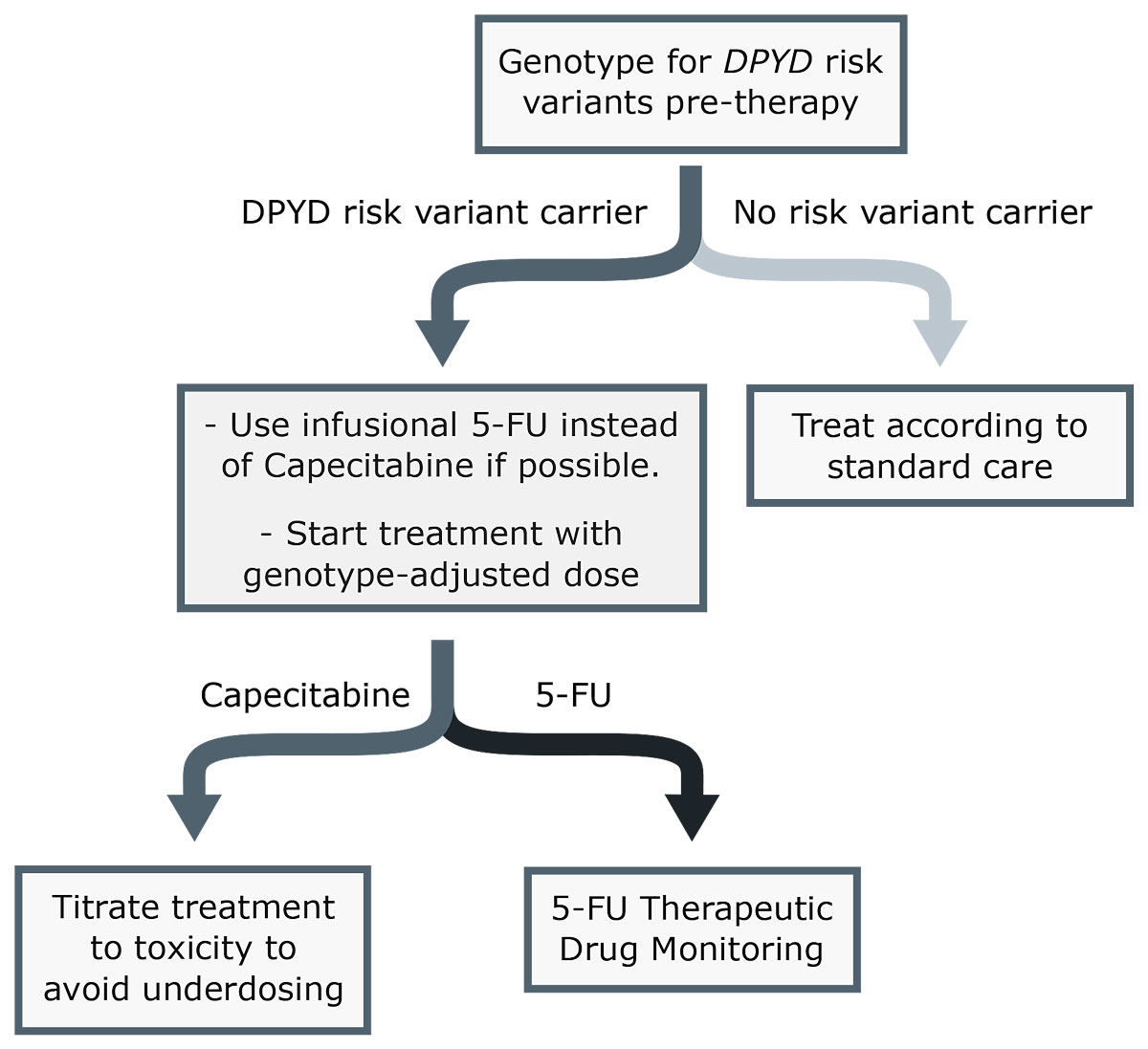
Fluoropyrimidine chemotherapy: recommendations for DPYD genotyping and therapeutic drug monitoring of the Swiss Group of Pharmacogenomics and Personalised Therapy | Swiss Medical Weekly

Teva Parenteral Medicines Initiates Voluntary Nationwide Recall of Select Lots of Adrucil® (fluorouracil injection, USP) 5 g/100 mL (50 mg/mL) Due to Particulate Matter | Business Wire