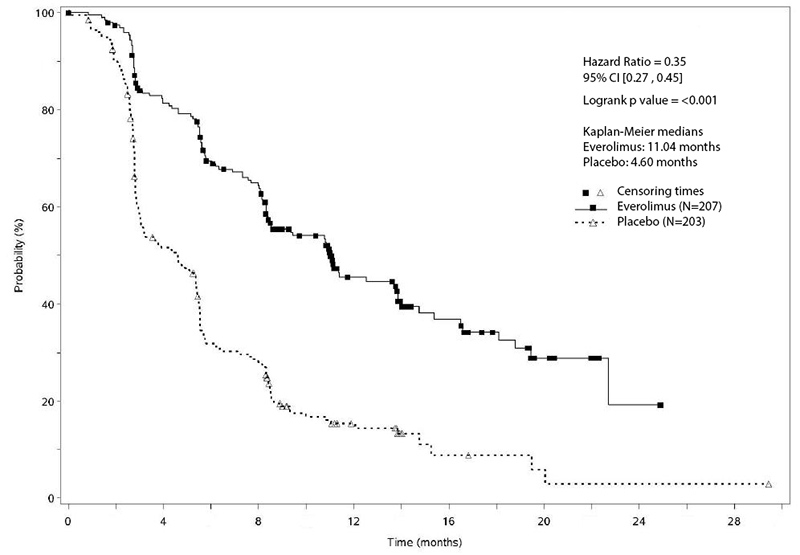
Randomized Phase II Trial of Fulvestrant Plus Everolimus or Placebo in Postmenopausal Women With Hormone Receptor–Positive, Human Epidermal Growth Factor Receptor 2–Negative Metastatic Breast Cancer Resistant to Aromatase Inhibitor Therapy: Results of
Bipacksedel: Information till patienten Everolimus Teva 2,5 mg Everolimus Teva 5 mg Everolimus Teva 7,5 mg Everolimus Teva 10 mg
These highlights do not include all the information needed to use EVEROLIMUS TABLETS safely and effectively. See full prescribing information for EVEROLIMUS TABLETS. EVEROLIMUS tablets, for oral use Initial U.S. Approval: 2009
These highlights do not include all the information needed to use EVEROLIMUS TABLETS safely and effectively. See full prescribing information for EVEROLIMUS TABLETS. EVEROLIMUS tablets, for oral use Initial U.S. Approval: 2009

Comparing everolimus‐based immunosuppression with reduction or withdrawal of calcineurin inhibitor reduction from 6 months after heart transplantation: The randomized MANDELA study - Barten - 2019 - American Journal of Transplantation - Wiley Online ...